Technology-driven mental health care with a human touch.
ANYWHERE. ANYTIME.
Innovative, digitally-enabled solutions for employee assistance, chronic condition management, managed behavioral health, and more.
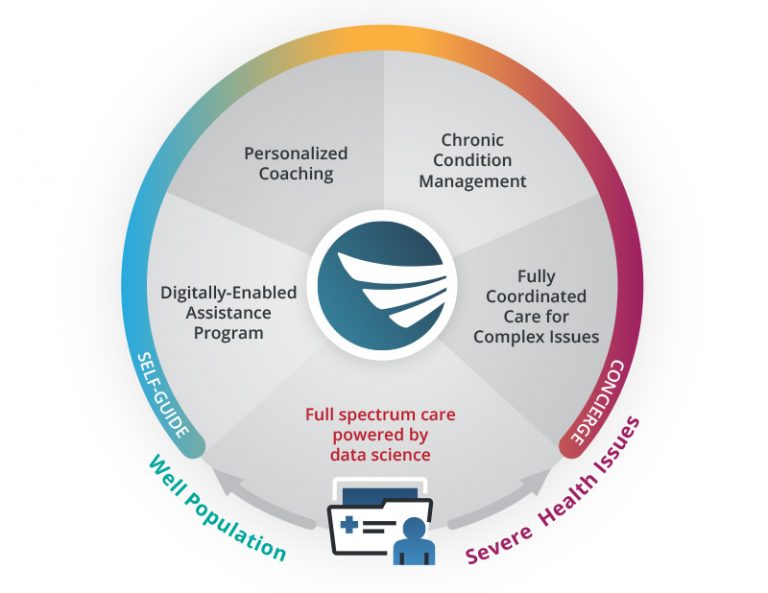
Improve access to employee mental health care with 4 EAP-related solutions.
More than 155 million Americans rely on employer-sponsored coverage for health care, and it benefits employers to make sure their employees are happy, healthy, and engaged. But barriers have been growing for decades, how can your organization respond?
A healthy mind and body at a fraction of the cost
Digitally-Enabled Assistance Program
With trainings, courses, 24/7 service, counselors, legal advice, and much more.Coaching and Counseling
Live coaches perform one-on-one sessions to guide members through issues.Chronic Condition Management
Coordinating care for chronic conditions like COPD, Cancer, and more.Managed Behavioral Health
Concierge level care for those with severe or complex health issues.
After rolling out digitally-enabled mental health solutions, Ōnin’s utilization rate increased by 161%.
Learn how The Ōnin Group prioritized employee mental health needs and saw dramatic improvements in utilization and engagement.
Trusted by the best













Helping organizations and people be their best for over 30 years
Uprise Health provides innovative technology-based tools and effective solutions for employee assistance, behavioral health, chronic condition management, wellness management, and substance abuse/opioid addiction. With a focus on positive outcomes through clinically validated methods and digital tools, we are able to reduce claims and costs for organizations while improving the lives of their members and families.





